 
Testing was conducted in a commercial diagnostic laboratory on day 1, when the first symptoms occurred. In all patients, SARS-CoV-2 RNA had been diagnosed via nasal and oro-pharyngeal fluids. Three days after patient 1, she began coughing, had elevated temperature (37–38 ☌) on days 2 and 3, muscle pain on day 3, rhinitis on days 5–7, and hyposmia on days 2–8. 
Subsequently, the patient recovered fully. Persistent tiredness started on day 4, anosmia occurred from day 5–13. Headache and limb pain were noted on days 2–8. On days 4 and 5, body temperature was elevated (37–38 ☌). The first symptom, coughing, occurred three days after the onset of disease in patient 1. Apart from occasional coughing after day 8, the patient’s well-being improved continuously until full recovery of health was reached. Symptoms on days 2–8 included headache, muscle pain in arms and legs, and fatigue. The first symptom was coughing, lasting until day 8 after onset of disease. By applying these test systems, the above-mentioned WHO standard was evaluated and used to measure the immune response of patients after a natural infection with SARS-CoV-2.Īll patients were related and lived in the same household. We have developed highly specific ELISA systems for the detection of IgG and IgA class antibodies directed to the spike (RBD) protein as well as to the nucleoprotein (N) of SARS-CoV-2. In addition, the effectiveness of the immune response after vaccination with the various established vaccines can now be compared. Hence, it is of utmost importance to define the humoral immune status of individuals based on measurements standardized with the now available WHO reference. Nevertheless, which quantities of antibodies reflect a valid protection against an infection with SARS-CoV-2 and/or against a COVID-19 disease are still poorly explored. The neutralizing activity of antibodies to SARS-CoV-2 is associated with IgG and IgA class antibodies directed against the receptor binding domain (RBD) of the spike protein. Thus, it is now possible to compare and harmonize different commercially available test systems as well as clinical and serological studies. In this standard preparation, the quantity of antibodies is defined as 1000 neutralization antibody units (IU) corresponding to 1000 binding antibody units (BAU) per mL. To solve this problem and to facilitate the comparison among different serological tests, the World Health Organization (WHO) has recently established and distributed reference material based on a standard serum pool collected from human donors having suffered from COVID-19. Until now, it has been difficult to compare the results obtained by different assay systems since the quantification of the viral ABs is not standardized. Most often, such assays measure antibodies of the classes IgG, IgA, and IgM. 
Usually, these ABs are directed against the SARS-CoV-2 surface S1-protein, the so-called spike protein, and the inner nucleocapsid protein N of the virus. So far, the detection of the humoral immune response after a SARS-CoV-2 infection has mainly been based on immunoassay systems measuring antibodies (ABs). Most likely, the wide use of the WHO reference preparation will be very useful in determining the individual immune status of patients after an infection with SARS-CoV-2 or after vaccination. Vaccination resulted in a rapid boost of antibodies to S1-protein but, as expected, not to the N-protein. Sera from all patients retained the ability to neutralize SARS-CoV-2 for more than a year. After natural infection, the antibodies (IgA, IgG) against the S1-protein remained elevated above the established cut-off to positivity (S-IgA 60 BAU/mL and S-IgG 50 BAU/mL, respectively) for over a year in all patients, while this was not the case for ABs against the N-protein (cut-off N-IgG 40 BAU/mL, N-IgA 256 BAU/mL). Furthermore, in two individuals, the effects of an additional vaccination with a mRNA vaccine containing the S1-RBD sequence on these antibodies were examined. Serum samples were collected frequently during a period of over one year. 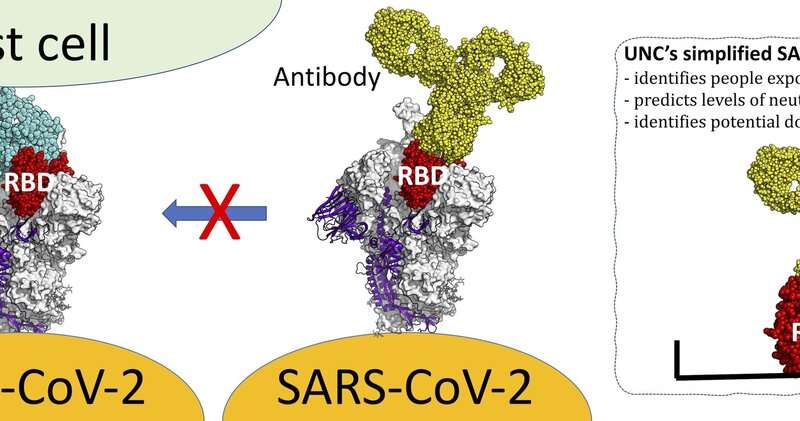
In this study, the concentration of antibodies (ABs) against both the S- and the N-protein of SARS-CoV-2 as well as serum neutralization activity were evaluated in three patients after a mild course of COVID-19. The new WHO reference standard allows for the definition of serum antibodies against various SARS-CoV-2 antigens in terms of binding antibody units (BAU/mL) and thus to compare the results of different ELISA systems. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
